PILA PHARMA’s TRPV1 asset
PILA PHARMA’s TRPV1 asset includes the clinical development candidate, XEN-D0501, back-up compounds and data.
The asset was previously owned by Ario Pharma/ Xention and, before that, Bayer Healthcare that invented the molecules and took XEN-D0501 through ‘first-in-man’ (as BAY69-9426 and BR-4874).
XEN-D0501 is a TRPV1 antagonist with a good clinical safety profile (as found in 300 exposed subject across 8 clinical trials). In patients with type 2 diabetes it was found to significantly improve insulin secretion.
PILA PHARMA is the owner of a use-patent giving the exclusive right to treat diabetes and obesity with TRPV1 antagonists (Read the use-patent application here)
In July 2022, the company received US Orphan Drug Designation for XEN-D0501 as treatment of the rare disease erythromelalgia.
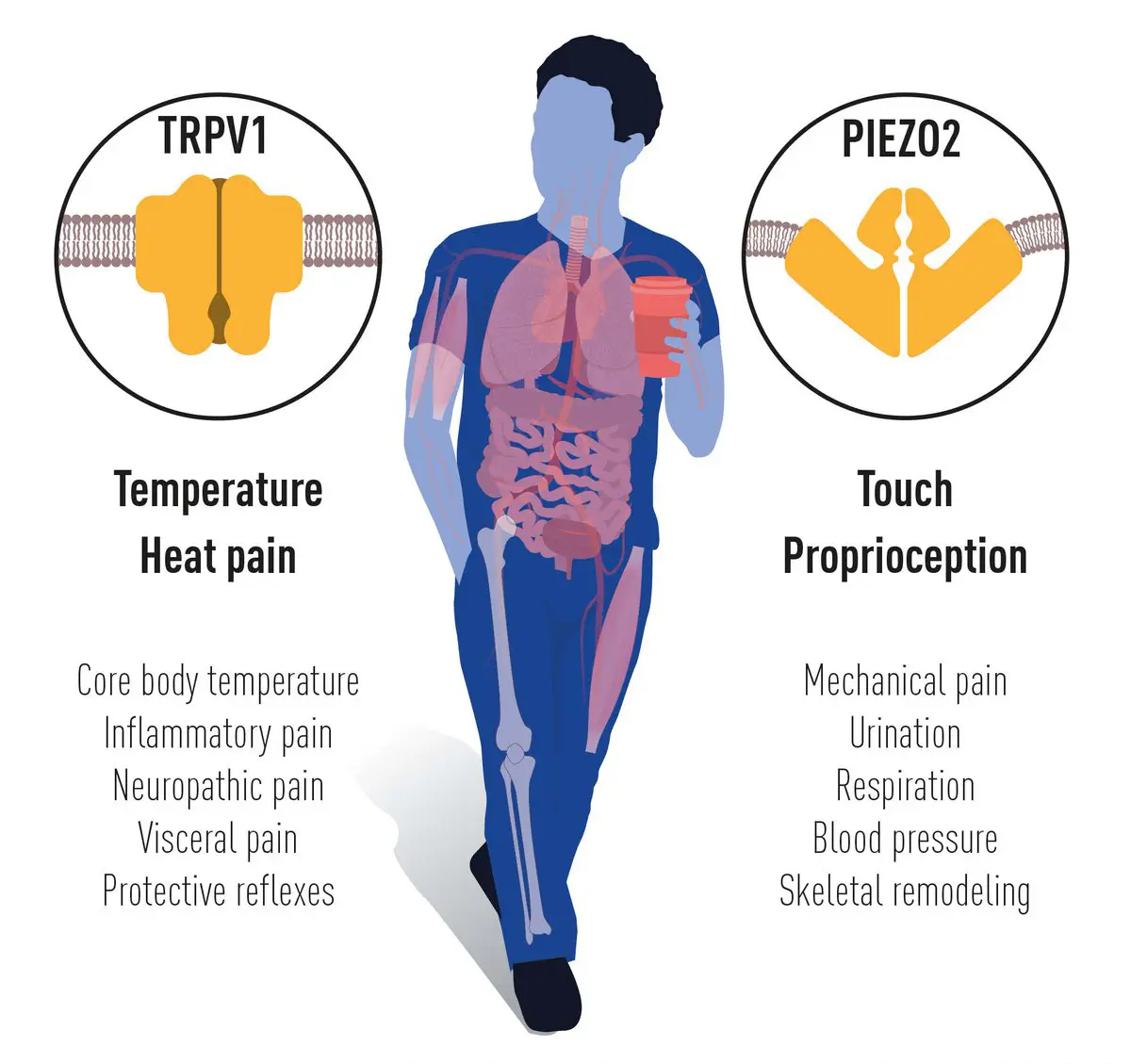
DISCOVERY OF TRPV1 AWARDED NOBEL PRIZE
IN 2021 DAVID JULIUS AND ARDEM WERE AWARDED THE NOBEL PRIZE IN PHYSIOLOGY OR MEDICINE FOR THEIR DISCOVERIES OF RECEPTORS FOR TEMPERATURE AND TOUCH.
DAVID JULIUS UTILIZED CAPSAICIN, A PUNGENT COMPOUND FROM CHILI PEPPERS THAT INDUCES A BURNING SENSATION, TO IDENTIFY A SENSOR IN THE NERVENDINGS OF THE SIKN THAT RESPONS TO HEAT,TRPV1.